| Hygroscopicity/Sorption behavior | [German version] |
| Definitions | |
| Sorption behavior of hygroscopic goods | |
| Types of sorption isotherm |
Definitions
| Hygroscopicity is the capacity of a product (e.g. cargo, packaging material) to react to the moisture content of the air by absorbing or releasing water vapor. Of decisive significance for the absorption or release of water vapor is the water content of a product. |
|
| The water content is the percentage of the total mass of a product constituted by water. |
|
| Sorption behavior describes the ability of a hygroscopic product to absorb or release water vapor from or into the air until a state of equilibrium is reached. |
|
| Adsorption means uptake of water vapor. |
|
| Desorption means water vapor release. |
|
| A sorption isotherm is the graphic representation of the sorption behavior of a substance. It represents the relationship between the water content of a product and the relative humidity of the ambient air (equilibrium) at a particular temperature. |
|
| Dry for shipment means that a product has a water content which does not cause it to suffer any depreciation under normal weather or hold/container conditions when cared for properly. |
|
| Critical water content means a water content which, if exceeded during transport or storage, may cause the onset of depreciative phenomena such as mold, fermentation, rot, self-heating/spontaneous combustion. If the water content falls below the critical value, desiccation losses may occur, such as fragmentation, drying-out, cracking. For safe transport, it is therefore important for the water content of a product to match the required values on acceptance and for this water content to be maintained in transit through the storage climate conditions in the hold/container. |
Back to beginning
Sorption behavior of hygroscopic products
The profile of a sorption isotherm is characteristic of the hygroscopicity of a product, the sorption behavior of a product being dependent on temperature. Highly hygroscopic products exhibit a steep sorption isotherm, while sparingly hygroscopic products exhibit flat sorption isotherms.
A measure of the hygroscopicity of a product is consequently the magnitude of the increase or decrease in its water content as a function of relative humidity at a certain temperature. Weakly hygroscopic products exhibit no or only a slight change in their water content as a consequence of variations in relative humidity. In strongly hygroscopic products, water content may vary widely.
By determining the water content of a product, it is possible, using a sorption isotherm, to establish how the product will behave in the hold/container.
1. The water content of a product is above the sorption isotherm:
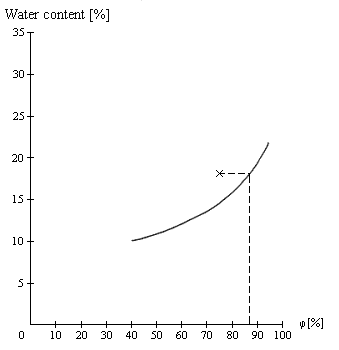
Figure 1
Figure 1 shows the sorption isotherm for wheat at 20°C. At a measured water content of 18% and relative humidity of the ambient air of 75%, the point of intersection lies above the sorption isotherm (see cross).
In an unventilated hold/container, the relative humidity would increase until a state of equilibrium is reached at a relative humidity of approx. 88% (at a water content of 18%). The mold growth threshold, which is at 75% relative humidity, is thus exceeded. Mold formation may proceed rapidly in an unventilated hold/container (the water content of the cargo remains high owing to the low level of water vapor release): this situation must be rectified by ventilation, the cargo theoretically being dried with suitable ventilation air until its water content is just below 15% and a state of equilibrium with the ambient air has therefore been reached. However, such thorough drying of moist cargo is not always possible in practice, and the cargo may spoil despite correct operation of the ventilation installation due to mold growth in areas of the hold or within the cargo which could not be reached by the ventilation air (dead air zones).
As an example, if 100 t of excessively wet grain with a water content of 16% has its water content reduced to 13%, 3 t of water vapor have to be removed by ventilation; if a hold contains 1000 t of grain, then the volume of water increases to 30 t. The capacity of the ventilation installation is inadequate for this purpose and the cargo is generally dried only superficially. This has the following consequences:
| the water content of the hygroscopic cargo must be checked by measurement prior to loading | |
| do not rely solely on shippers‘ certificates | |
| if appropriate, commission an inspector | |
| do not accept goods with an excessively high water content or at least make an appropriate note in the accompanying documents, so that, in the event of subsequent sweat damage, the excessive water content at the time of acceptance of the cargo may be acknowledged as a cause. |
2. The water content of a product is below the sorption isotherm:
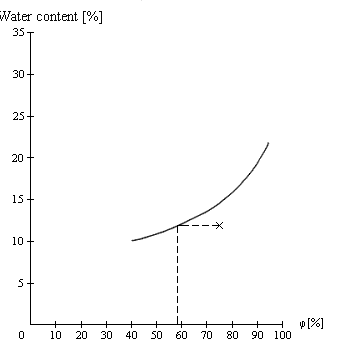
Figure 2
Figure 2 shows the sorption isotherm for wheat at 20°C. At a measured water content of 12% and a relative humidity of the ambient air of 75%, the point of intersection lies below the sorption isotherm (see cross).
In an unventilated hold, relative humidity would decrease as a result of the absorption of water vapor by the hygroscopic product until a state of equilibrium is reached at a relative humidity of just below 60%. Since the amount of water vapor available for absorption is small, the water content of a product hardly changes during this process. In general, there is no risk of depreciation and ventilation is unnecessary, unless the product is prone to desiccation damage (tobacco, tea, wood, leather). In this case, the hold/container would have to be ventilated with suitable ventilation air to prevent drying-out.
To summarize:
| In an unventilated hold/container filled with a hygroscopic product, the product determines the relative humidity in the hold, i.e. the product creates its own atmospheric environment. During these balancing processes, the product itself undergoes only slight changes to its water content, since this quantity of water is a multiple of the total (absolute) humidity of the hold/container air. | |
| In a ventilated hold/container filled with a hygroscopic product, the relative humidity of the hold/container depends on the external air values, the product being able to absorb or release water vapor accordingly. It should be noted that only when a product is subjected to constant ventilation with excessively moist or dry air for several days or weeks does its water content increase or decrease slowly. It should also be noted that, even when a hold/container is ventilated, the air between particles (e.g. cereal grains, coffee or cocoa beans) or in dead air zones behaves as if the hold/container were closed, i.e. unventilated. | |
| Influence of cargo temperature on equilibrium moisture content: any changes in product temperature result in different equilibrium moisture conditions. Since, for example, a 10°C sorption isotherm is higher than a 20° sorption isotherm, identical water contents correspond to lower air equilibrium moisture contents, e.g. while an equilibrium moisture content of approx. 75% applies to wheat with a water content of 15% and an intrinsic temperature of 20°C, when the intrinsic temperature is 10°C, the equilibrium moisture content is approx. 68%. | |
| Consequently, on a North Atlantic crossing the admissible water content for grain may be higher at the prevailing low air temperatures without the critical relative humidity > 75% (mold growth threshold) being exceeded in the hold/container. | |
| For vegetable products loaded in tropical ports, on the other hand, this means that it is essential to take full advantage of every opportunity to cool the goods, so that the lowest possible relative humidity prevails in the hold/container should it be necessary to switch off the ventilation. Warm products release greater amounts of water vapor and lead more quickly to sweat formation. |
Back to beginning
Types of sorption isotherm
In general, a distinction may be drawn between three types of sorption isotherm:
1. Sorption isotherm rises steeply, i.e. the product is strongly hygroscopic, as is the case with dried fruits and desiccants for corrosion protection.
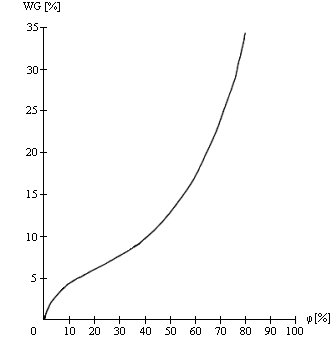
Figure 3
2. Sorption isotherm exhibits an S-shaped profile, its central portion constituting the range in which properties are at their most stable, i.e. over this range no harmful changes are to be expected in the product, such as foodstuffs, paper etc..
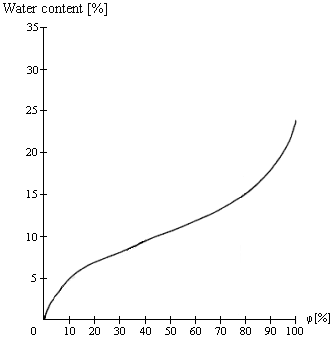
Figure 4
3. Over a wide range, the product exhibits low hygroscopicity (anhydrous form); once the flow moisture point has been reached, the product rapidly absorbs large amounts of water vapor, causing it to deliquesce (hydrous form), as in the case of many crystalline products (salt, sugar, potash, tartaric acid).
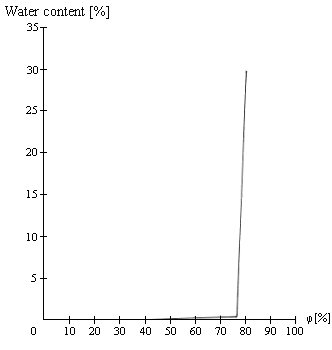
Figure 5
Back to beginning
